According to China Center for Drug Evaluation (CDE)'s website, Sanofi's avalglucosidase alfa entered the preliminary list of priority review designation on August 15. If no objections are filed to CDE by August 22, the drug will officially be granted the priority review.
Avalglucosidase alfa is indicated for long-term enzyme replacement treatment for Pompe disease, a rare disease with an incidence rate of 1/50,000 in China.1 The disease is caused by the genetic deficiency or dysfunction of the lysosomal enzyme acid α-glucosidase (GAA), resulting in glycogen accumulation in muscles, including the proximal muscles and the diaphragm, and eventually causing progressive and irreversible muscle damage.2
China encourages the development of rare disease drugs by allowing them to apply for priority review designation. If officially granted with the priority review, avalglucosidase alfa can complete the technical review within 70 workdays, much shorter than 200 workdays for drugs without the priority review.
Related Articles:
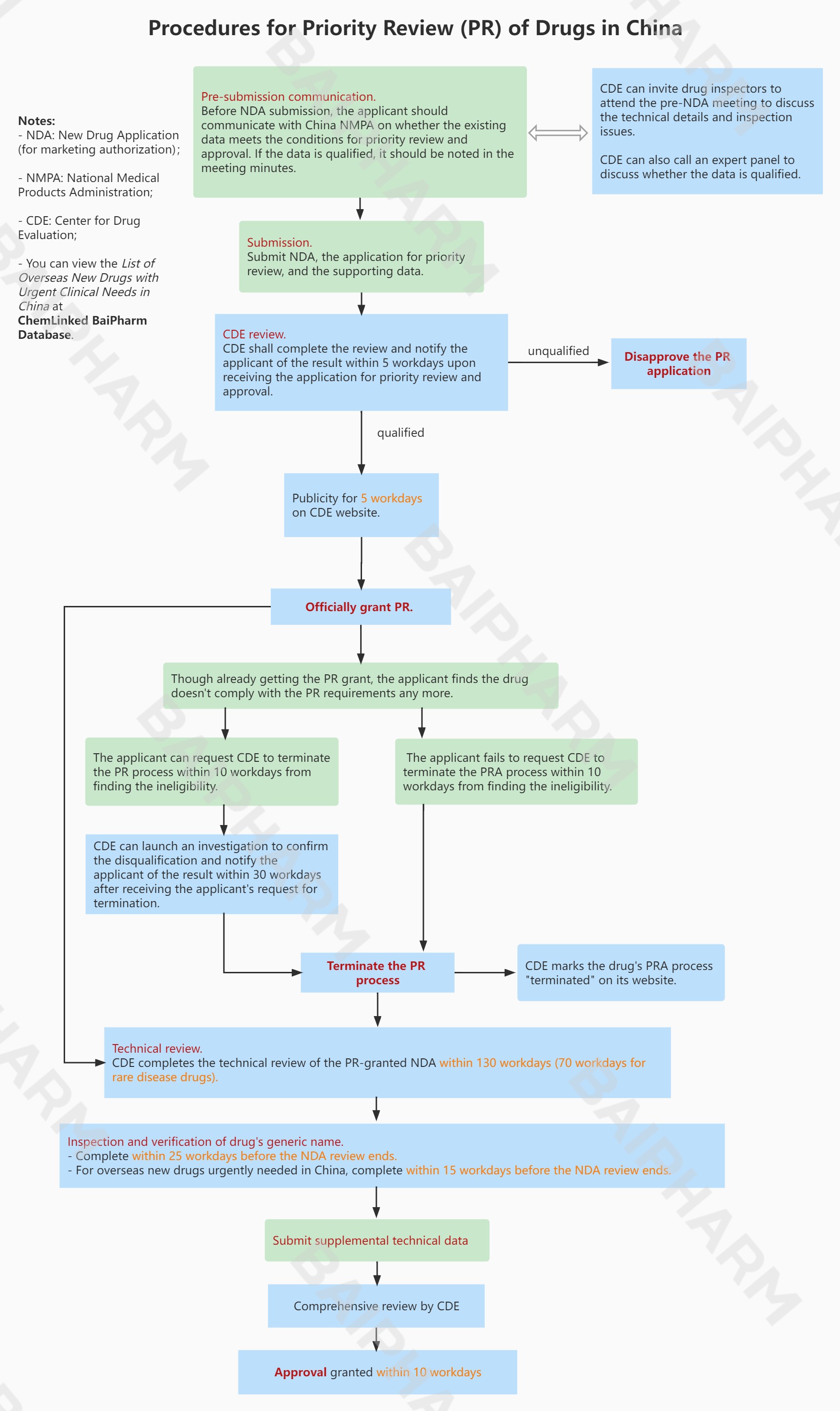 Procedures for Priority Review in China
Procedures for Priority Review in China
List of 27 Drugs With Priority Review Designation
Since the beginning of 2022, China has granted priority review designation to 27 drugs, as shown below.
No. | Drug Name or Code | Applicant | Publicity Date | Indication(s) for Priority Review |
1 | Triptorelin Pamoate for Injection | Ipsen Pharma | 2022/7/21 | For treating: Locally advanced and metastatic prostate cancer; Endometriosis (stages I-IV); Central precocious puberty (girls ≤ 8 years old and boys ≤ 10 years old) |
2 | Methylphenidate Hydrochloride Prolonged-release Chewable Tablets | Pediatrix Therapeutics | 2022/7/19 | For treating attention deficit hyperactivity disorder (ADHD) |
3 | Olverembatinib Tablets | Guangzhou Shunjiian Biomedical Technology | 2022/7/11 | For treating patients with chronic myeloid leukemia (CML) patients in the chronic phase who are resistant and/or intolerant to first- and second-generation tyrosine kinase inhibitors (TKIs) |
4 | Crovalimab Injection | Roche | 2022/7/11 | For treating paroxysmal nocturnal hemoglobinuria in adults and adolescents (≥12 years old) who are currently not receiving complement inhibitor therapy |
5 | Narlumosbart for Injection | JMT Biotech | 2022/7/8 | For treating giant cell tumor of bone that is unresectable or difficult to be operated by surgery |
6 | Phentolamine Mesylate Injection | Chengdu Xinjie High-tech Development | 2022/6/16 | For controlling paroxysmal hypertension in patients with pheochromocytoma before and during surgery; For the diagnostic test of pheochromocytoma if other specific tests cannot be performed or when other tests are not sufficient to give a definitive diagnosis; For preventing skin necrosis and decay caused by norepinephrine spillage. |
7 | IBI326 | IASO Biotherapeutics | 2022/6/2 | For treating adult patients with relapsed or refractory multiple myeloma after third-line or more systemic therapy |
8 | Selumetinib Capsules | AstraZeneca | 2022/5/25 | For treating neurofibromatosis type 1 (NF1) in pediatric patients no younger than 3 years old with symptomatic and inoperable plexiform neurofibromatosis (PN) |
9 | Proglycem Oral Suspension | Suzhou E-fan Pharmaceuticals | 2022/5/23 | For treating hypoglycemia due to hyperinsulinemia |
10 | Digoxin Injection | Brilliant Pharmaceuticals | 2022/5/12 | For increasing left ventricular ejection fraction in adult patients with mild to moderate heart failure. The drug can improve symptom of heart failure, as manifested in stronger exercise capability and fewer heart failure-related hospitalizations and emergency treatments, but has no effect on mortality. When possible, the drug should be used in combination with diuretics and angiotensin-converting enzyme (ACE) inhibitors. For increasing myocardial contractility in pediatric heart failure patients. For controlling ventricular response rate in adult patients with chronic atrial fibrillation. |
11 | Clobazam Tablets | Yichang Humanwell Pharmaceutical | 2022/5/11 | Used in combination therapy for treating seizures in patients with Lennox-Gastaut syndrome (LGS) who are no younger than 2 years old. |
12 | Sodium Thiosulfate Injection | Huiyu Pharmaceutical | 2022/5/11 | Mainly for treating cyanide poisoning; Also for treating arsenic, mercury, lead, bismuth, and iodine poisonings |
13 | Stiripentol Dry Suspension | SJZ No.4 Pharmaceutical | 2022/5/11 | Used as an adjuvant therapy, in combination with clobazam and valproate, for treating seizures in infant patients with refractory severe myoclonic epilepsy (Dravet syndrome, also known as SMEI) that cannot be sufficiently controlled by clobazam and valproate. |
14 | Methotrexate Injection (Pre-filled) | Shenzhen Kangzhe Biotech | 2022/5/11 | For treating severe, intractable, disabling psoriasis that is insensitive to conventional treatments. |
15 | Carglumic Acid Dispersible Tablets | Recordati | 2022/5/11 | For treating adults and pediatric patients with hyperammonemia caused by primary deficiency, isovaleric acidemia (IVA), methylmalonic acidemia (MMA), and/or propionic acidemia (PA). |
16 | Cytarabine for Injection | Guoyao Yixin Pharmaceutical | 2022/4/24 | Mainly for the induction and maintenance treatment of acute non-lymphocytic leukemia in adults and pediatric patients. |
17 | Trastuzumab Deruxtecan for Injection | Daiichi Sankyo | 2022/4/24 | As a single agent for treating unresectable or metastatic HER2-positive breast cancer in adult patients who have received one or more anti-HER2 drugs. |
18 | Shingles Vaccine, Live | Changchun BCHT Biotechnology | 2022/4/7 | For preventing shingles |
19 | Relmacabtagene Autoleucel Injection | JW Therapeutics | 2022/3/24 | For treating adult patients who have received second-line or more systemic therapy for relapsed or refractory follicular lymphoma, including follicular lymphoma with the histological grade 1, 2, or 3a |
20 | Sucroferric Oxyhydroxide Chewable Tablets | Hangzhou Tigermed | 2022/3/24 | For treating hyperphosphatemia in adult patients with chronic kidney disease (CKD) who are receiving hemodialysis (HD) or peritoneal dialysis (PD); For treating hyperphosphatemia in pediatric CKD patients no younger than 12 years old who have stage 4-5 CKD (glomerular filtration rate < 30 mL/min/1.73 m2) or are receiving dialysis |
21 | Influenza Vaccine (Split Virion), Inactivated,Quadrivalent | Shanghai Institute of Biological Products | 2022/3/17 | After inoculation, the vaccine can stimulate antibodies to produce immunity against influenza virus. The vaccine is indicated for preventing influenza caused by vaccine-related types of influenza viruses. |
22 | Perampanel Oral Suspension | Eisai | 2022/3/17 | For treating partial seizures (with or without secondary generalized seizures) in adults and children no younger than 4 years old |
23 | Empagliflozin Tablets | Boehringer Ingelheim | 2022/3/2 | For reducing the risk of cardiovascular death and hospitalization due to heart failure in adult patients. The clinical benefit is irrelevant to left ventricular ejection fraction and is evident in adults with reduced, intermediate, or preserved ejection fraction. |
24 | Spesolimab Injection | Boehringer Ingelheim | 2022/3/1 | For treating generalized pustular psoriasis (GPP) |
25 | Glumetinib Tablets | Haihe Bipharma | 2022/2/17 | For treating locally advanced or metastatic non-small cell lung cancer (NSCLC) with MET14 exon skipping |
26 | Influenza Vaccine (Split Virion), Inactivated, Quadrivalent | Shenzhen Sanofi Pasteur Biological Products | 2022/1/10 | For preventing influenza in people at the age between 6-35 months, especially people susceptible to influenza-related complications |
27 | Maralixibat Oral Solution | CANbridge Life Sciences | 2022/1/10 | For treating cholestatic pruritus in patients no younger than 1 years old with Alagille syndrome |
Find out more about priority review designation at 2021 China CDE Drug Evaluation Report-4.3 Priority Review (PR)










