According to China's Administrative Measures for Drug Registration enacted since July 1, 2020, the National Medical Products Administration (NMPA) shall launch expedited programs for drug registration to support clinical-value-oriented innovation in drugs.1
China currently follows four programs for accelerating the review and approval of new drug applications (NDA) that meet the corresponding criteria.
breakthrough therapy designation (BTD);
conditional approval (CA);
priority review (PR);
special approval (SA).
If an NDA enter any of the above programs, NMPA and its professional institutions will offer policy and technology support to the applicant. The support includes necessary technical guidance, communication with the applicant, preferential allocation of resources, and shortening the review time.
1. Regulatory Updates on Drug Review and Approval in China
To take the lead in the ever-changing Chinese market, international pharmaceutical companies need to keep abreast of the regulatory updates of marketing authorization procedures:
Table 1: Laws, Regulations and Policies Involving Requirements for Drug Review and Approval in China
Issued/ Revised | Enforced | Law/regulation/policy |
08/10/2017 | 08/10/2017 | Opinions on Deepening the Reform of Review and Approval System to Encourage Innovation in Drugs and Medical Devices |
26/08/2019 | 01/12/2019 | |
02/03/2019 | 02/03/2019 | Provisions on Implementing the Drug Administration Law |
15/01/2020 | 01/07/2020 | Administrative Measures for Drug Registration |
08/07/2020 | 08/07/2020 | Work Process for Breakthrough Therapy Designation of Drugs (Trial); Work Process for Conditional Approval of Drugs (Trial); Work Process for Priority Review of Drugs (Trial)2. |
19/11/2020 | 19/11/2020 | Technical Guideline for Drugs Applying for Conditional Marketing Authorization (Trial) |
2. For International Companies: Recommended Process for Starting Drug Marketing Application in China
① Choose the correct registration classification for the drug product;
② Analyze and evaluate the overseas clinical data, see if it can be accepted by China Center for Drug Evaluation (CDE), and if the application would be qualified for an expedited program for drug registration;
③ Find a legal agent in China to conduct data gap analysis;
④ Propose a pre-submission meeting or written communication with CDE;
⑤ Submit the Investigational New Drug (IND) application or file a record of Bioequivalence (BE) study for generic drugs to CDE;
⑥ Submit marketing authorization application for imported drugs after completing relevant researches.
3. Four Expedited Programs for Drug Registration in China
3.1 Breakthrough Therapy Designation (BTD)
3.1.1 Reference
Work Process for Breakthrough Therapy Designation of Drugs2 (effective since July 8, 2020);
Administrative Measures for Drug Registration (effective since July 1, 2020);
Administrative Rules of Consulting the Common Technical Issues on Reviewing Drug Registration Application3 (effective since Dec. 21, 2018);
Administrative Measures for Communication on Drug Development and Technical Review4 (effective since Oct. 8, 2018).
3.1.2 Criteria
The product should be an innovative or improved new drug that meet both conditions below:
① For a life-threatening disease or disease that seriously impacts life quality;
② The product uses a new effective therapeutic method for the disease stipulated in ① ; or with sufficient evidence showing substantial clinical improvement over the existing therapy.
Notes on three concepts in the expedited program:
Life-threatening conditions:
Refer to severe, incurable or irreversible conditions which lead to shortening of life or death.
Conditions seriously impacting life quality:
Refer to severe conditions that have substantial impact on normal physiologic function and will progress to disability or deficiency of significant physiological and social functions without effective treatment.
Existing therapy:
Refer to a therapy that has already approved or licensed in China for the same indication of the new drug; or the therapy is a standard of care (SOC) such as drug-device combination therapy.
3.1.3 When to Submit Request
In phase I or phase II of the clinical trial, generally not later than the beginning of the phase III of the clinical trial.
3.1.4 CDE's Preferential Policy
An applicant can propose communication with CDE during the pivotal stage of during the clinical trials.
An applicant can submit phased research data to CDE, which will then give advice or comments on the research plan and send a feedback to the applicant.
3.1.5 BTD Application Procedures
 Procedures for Breakthrough Therapy Designation (BTD) of Drugs in China
Procedures for Breakthrough Therapy Designation (BTD) of Drugs in China
3.2 Conditional Approval (CA)
3.2.1 Reference
Technical Guidance for Drugs Going to the Market with Conditional Approval (Trial)5 (effective since Nov. 19, 2020)
Work Process for Conditional Approval of Drugs (Trial) (effective since July 8, 2020);
Measures for Drug Registration (effective since July 1, 2020).
3.2.2 Criteria
① Drug for serious life-threatening conditions which currently have no effective treatment; and the drug’s existing clinical data have demonstrated the drug's effectiveness and supported its clinical value.
② Drug urgently needed for public health; and the drug’s existing clinical data have demonstrated the drug's effectiveness and supported its clinical value.
③ Vaccines that are recognized by the NHC that they are urgently needed for severe public health emergencies, or for other urgent needs; and as evaluated, benefits outweigh risks.
3.2.3 CDE's Preferential Policy
Grant the drug marketing approval even before the drug research completes, and allow some of the research to be completed in allocated time after marketing.
3.2.4 CA Application Procedures
 Procedures for Conditional Approval (CA) of Drugs in China
Procedures for Conditional Approval (CA) of Drugs in China
3.3 Priority Review (PR)
3.3.1 Reference
Work Process for Priority Review of Drugs (Trial) (effective since July 8, 2020);
Measures for Drug Registration (effective since July 1, 2020).
3.3.2 Criteria
During the marketing authorization application stage, the drug with high clinical value shall meet one of the following conditions:
① Drugs in shortage and with urgent clinical needs, or are innovative/improved new drugs to prevent and treat serious infectious/rare diseases;
② New drugs or drugs with new dosage forms and strengths for pediatrics;
③ Vaccines with urgent needs for preventing and treating diseases or innovative vaccines;
④ Drugs granted Breakthrough Therapy Designation.
⑤ Drugs eligible for Conditional Approval.
⑥ Other situations specified by National Medical Products Administration (NMPA).
3.3.3 CDE's Preferential Policy
Accelerate technical review;
If necessary, prioritize the verification of the drug's generic name;
Accelerate administrative review.
3.3.4 PR Application Procedures
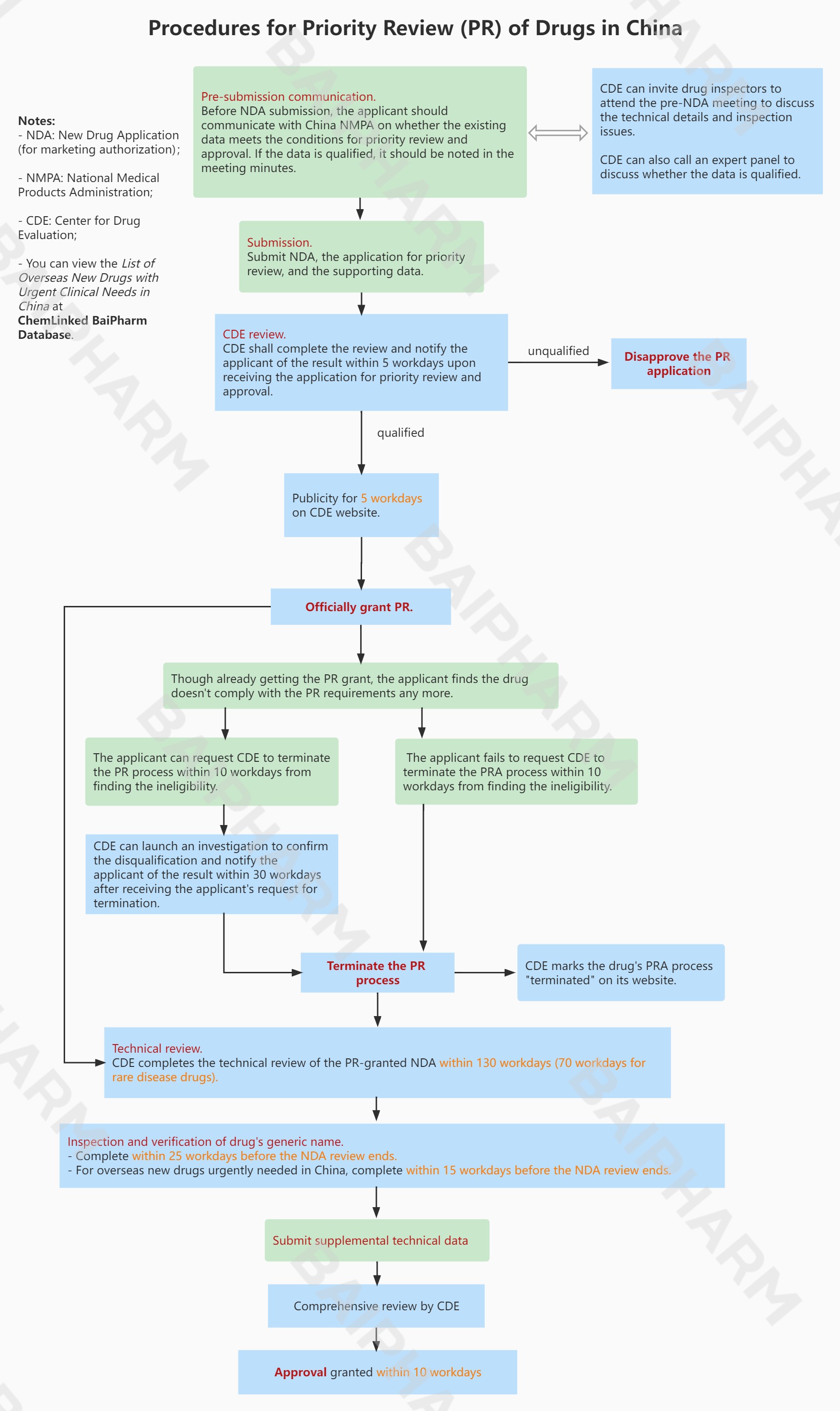 Procedures for Priority Review of Drugs in ChinaYou can view the List of Overseas New Drugs with Urgent Clinical Needs in China in ChemLinked BaiPharm Database.
Procedures for Priority Review of Drugs in ChinaYou can view the List of Overseas New Drugs with Urgent Clinical Needs in China in ChemLinked BaiPharm Database.
3.4 Special Approval (SA)
3.4.1 Reference
Measures for Drug Registration (effective since July 1, 2020);
Work Process for Special Approval of Drugs (effective since Nov. 18, 2005)
3.4.2 Criteria
When threatened by the ongoing public health breaking event or after an emergency, NMPA can legally conduct special approval of the prophylactic and therapeutic drugs needed in an emergency. The drugs that obtained special approval can be used in a limited time and area according to the particular needs for disease prevention and control.
3.4.3 SA Application Procedures
 Procedures for Special Approval (SA) of Drugs in China
Procedures for Special Approval (SA) of Drugs in China
Leave a message at BaiPharm contact box for reliable answers and solutions for drug registration.











