China's pharmaceutical industry has experienced rapid progress since the country entered the World Trade Organization. As of 2021, China holds 12% of the global pharmaceutical market, following the United States, responsible for 40% of the total revenue worldwide. According to Xinhua, the Chinese pharmaceutical market generated 708.75 billion yuan in 2021, while Chinese domestic pharmaceutical companies amassed 502 billion USD (3.37 trillion RMB) in revenue.
The immense growth of China's healthcare industry has allowed it to begin the transition from being a pharmaceutical manufacturing base to a strategic R&D hub. As a result, China has cemented itself as a strategic player in the global pharmaceutical market, both as a consumer country and as a platform for establishing greater R&D. The local pharmaceutical sector is pushed to grow with government incentives and public insurance but is hindered by IP laws and tedious regulations.
The Government's Efforts in Bolstering China's Pharmaceutical Industry Domestically and Abroad
Currently, China is facing the problem of promoting the growth of its pharmaceutical industry while ensuring affordable and easily accessible drugs for its patient population. In tackling this issue, the central government has emphasized a top-down approach. China's healthcare expenditure in relation to its GDP was 7.1% in 2020. In addition to investing heavily, the government has rolled out a series of broad changes to the pharmaceutical regulatory environment. The policy agendas outlined in China's 14th Five-Year Plan and Healthy China 2030 initiative each reinforce the importance of healthcare reforms such as decreasing inequality in the insurance coverage ranges and strengthening public services. Reform measures within the industry also aim at increasing access to medicine, making medicine affordable, and developing silver economy products for China's rapidly aging population.
Pharmaceutical policy in China has historically been relatively disjointed and uncoordinated between provinces, but these initiatives show that China is working towards restructuring the industry with the objective of improving its ability to compete internationally while still ensuring the benefits that come with the expansion are passed to domestic patients.
Along these lines, as part of the "Made in China 2025" industrial plan, China hopes to reinvent its pharmaceutical industry. The pharmaceutical industry is one with high technological benchmarks that require massive amounts of research and development (R&D). However, with such high investment costs, most Chinese companies are simply priced out. Beijing's ultimate goal for the domestic pharmaceutical industry is to create national champion companies that can dominate the Chinese market, compete and beat foreign competitors, and begin to take market share abroad.
China's Pharmaceutical Policy in the 14th Five-Year Plan
The 14th Five-Year Plan, in comparison to the 13th Five-Year Plan, shifts from calling on Chinese pharmaceutical producers to "follow innovation" to "becoming leaders in innovation". Similarly, an aim of reaching an average annual increase of more than 10% in R&D investment across the industry was set as a target, and investors are encouraged to invest in Chinese pharmaceutical R&D. Along with the transformation of the vocational education sector, pharmaceutical and biomedicine talent cultivation was put on the agenda.
In view of the international pharmaceutical market, the 14th Five-Year Plan stipulates increasing cooperation with overseas research institutions and drug companies. This is to be achieved through research cooperation abroad but also by creating better conditions for MNCs to set up innovative pharmaceutical production sites in China.
Historical Development of China's Pharmaceutical Manufacturing Industry
As China pursued policies of self-sufficiency from the 1940s to the 1980s, the country developed its pharmaceutical industry to meet local needs. It established large industrial zones and improved infrastructure to support the pharmaceutical manufacturing industry. It had its own standards and frameworks in terms of drug development that were very different from international standards. Since joining the WTO in 2001, China had to change many of these policies and regulatory frameworks. However, many of the largest domestic pharmaceutical companies in China are mostly state-owned, but with shares listed on public securities exchanges, i.e. mixed public and private ownership.
China's pharmaceutical manufacturing industry developed when the country was relatively isolated from international trade. In particular, the pharmaceutical manufacturing industry manufactures chemical, medicinal, and pharmaceutical products in various formats, including ampules, tablets, capsules, vials, ointments, powders, solutions, suspensions, and radioactive medicine. These products are then sold through pharmacies or used in hospitals. China's pharmaceutical manufacturing sector supplied the needs of the country's population and grew rapidly as the nation transitioned from a centrally planned economy to a market-based economy. Thus, it is evident that the pharmaceutical manufacturing industry is an important part of China's national economy.
In 2021, China has over 5,000 pharmaceutical manufacturers, and the Government is strongly promoting the consolidation of the pharmaceutical industry, especially to reduce the number of smaller companies that do not have the financial capacity to meet new regulatory standards.
Chinese pharmaceutical manufacturers have largely focused on the production of basic chemicals, intermediates, and active pharmaceutical ingredients (APIs), with roughly 4,000 API manufacturers in the country. In a relatively short period of time, China has become the leading global supplier of APIs in terms of volume. More recently, Chinese manufacturers have focused on the development and production of finished pharmaceutical products (FPPs), primarily to serve the domestic market, but with increasing attention to export markets.
The statistic below shows a breakdown of the prime operating revenue of China's pharmaceutical manufacturing industry in 2018, by segment. In 2018, the total revenue of the pharmaceutical sector in China amounted to 2.6 trillion yuan.
 Source: Statista, China MIIT. Designed by daxue consulting. Revenue Breakdown of the Pharmaceutical Industry in China 2018
Source: Statista, China MIIT. Designed by daxue consulting. Revenue Breakdown of the Pharmaceutical Industry in China 2018
Current State of China's Pharmaceutical Market
Chinese pharmaceutical companies are primarily engaged in the production of generics, therapeutic medicines, active pharmaceutical ingredients, and traditional Chinese medicine. More recently, the industry switched from focusing solely on creating generic drugs to creating more innovative drugs. As of 2022, 95% of all drugs approved by the National Medical Products Administration were still generics.
China's pharmaceutical market is also highly fragmented – a majority of manufacturers are small and medium-sized companies. Thus R&D spending is low compared with global competitors. The 14th Five-Year Plan aims to increase spending to 10% of sales by 2025. Pharmaceutical MNCs such as AstraZeneca, Novartis, and Novo Nordisk have strong market positions, each holding between 8-10% of the domestic market share in 2019. The market is highly dependent on distributor networks, leading domestic pharmaceutic players including Sinopharm and Shanghai Pharmaceuticals to have strong control. As a result of the industry's fragmentation, the government has been seeking to consolidate the pharmaceutical sector and increase the average size of firms to support quality inspection and improvement.
Aside from the aforementioned policy changes, major medical reforms have been enacted to boost China's pharmaceutical industry's maturity and growth. These reforms aim to tackle stringent drug quality regulations, incentivize medical innovation, and reduce intermediaries within the pharmaceutical distribution sector. Moreover, the National Medical Products Administration (NMPA) have adjusted its testing requirements in 2015 regarding the quality of off-patent generic drugs to lift industry standards and bring to speed foreign drug reviews.
China's pharmaceutical sector has some advantages over foreign countries when it comes to producing drugs. Due to its population size, sufficient numbers of patients for testing vaccinations on rare diseases can be recruited for testing in China, compared to the US and other western countries. This in turn produces a greater quantity of clinical data and boosts the development of ground-breaking drugs in China. However, clinical data is held and sold to pharmaceutical companies on a provincial level, which makes it harder for international pharmaceutic companies to obtain.
Additionally, to cut excessive drug price markups, China rolled out a volume-based procurement mechanism for hospital tenders in 2020 to encourage off-patent generics makers to out-price originators. According to China Health Insurance Research Association, in the first five procurement rounds, bidding prices of expired patent originator drugs dropped on average by over 43% and original producers only won 10 to 32% of all tenders.
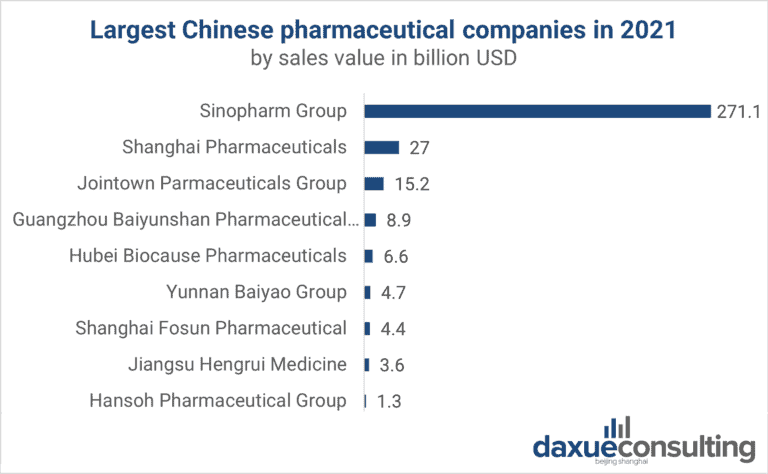 Source: Forbes Global 2000, Statista, the largest Chinese pharmaceutical companies with a sales value above 1 billion US Dollars as of May 2021
Source: Forbes Global 2000, Statista, the largest Chinese pharmaceutical companies with a sales value above 1 billion US Dollars as of May 2021
China's Pharmaceutical Market Size
In 2018, the market size of pharmaceutical and health products in China reached 220 billion USD, while 29.4% of these products were supplied by imports. By 2022, China's pharmaceutical market is estimated to reach 573.5 billion USD.
The Chinese pharmaceutical market segments include generics, over-the-counter (OTC) medicines, and patented medicines. Due to the low purchasing power and a robust local manufacturing sector, generic drugs (85.3 billion USD in 2019) seized a large share of China's pharmaceutical market. Sales of OTC medicines (18.4 billion USD), which represent 13.6% of the market are supported by a cultural acceptance of self-medication and fairly liberal sales channels. Patented medicines (30 billion USD), which are primarily consumed by China's affluent class contributed a small portion (22.9%) to overall pharmaceutical sales.
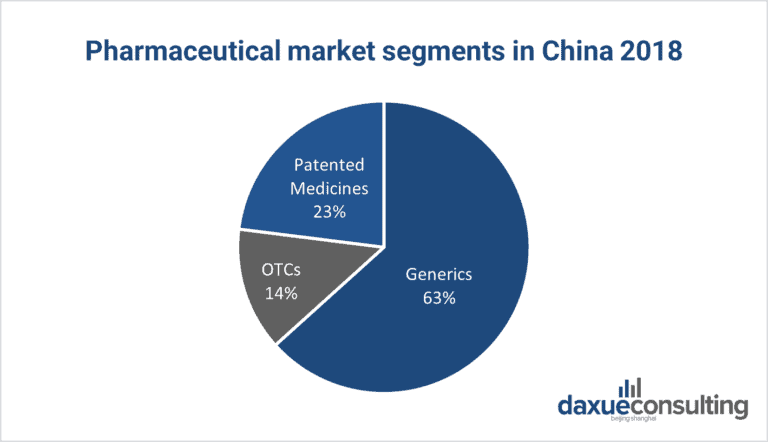 Source: U.S. Department of Commerce. Designed by daxue consulting. Pharmaceutical market segments in China 2018
Source: U.S. Department of Commerce. Designed by daxue consulting. Pharmaceutical market segments in China 2018
Although China has expanded to be the second largest pharmaceutical market worldwide, growth has fluctuated from a CAGR of 19% and 8% between 2008 and 2013 but is expected to reach 12.2% by 2025. Much of China's growth over the past ten years has been driven by central government reforms to expand insurance access to both rural and urban residents, expand and modernize the hospital system, and reform to better integrate primary care services. Growth moving forward will be attributed to the improvement of treatment concepts, the optimization of medical measures, the acceleration of new drug access, the improvement of medical service quality, and the dynamic adjustment of medical insurance access.
Key Products in China's Pharmaceutical Industry
Generic Medicine in China
This includes anti-infection drugs (the leading generics segment), cardiovascular system drugs (second largest market with the highest ROI), anti-tumor and cancer drugs, and drugs for the digestive system.
Generic drugs are the mainstay of China's pharmaceutical industry. Due to its large market demand, this segment's revenue is expected to increase strongly in future years, as China will utilize generics in its public insurance plan to minimize overall healthcare expenditures.
Patent Medicine in China
Patent medicine is the second-largest product segment, with high-profit margins. However, high R&D costs, long R&D periods, and long return periods are involved in the production of originator drugs. State policy aims to increase the share of innovative domestically created originator drugs, but rapid policy changes may pose a threat in terms of long-term profitability. As of 2022, foreign companies are still the main participants in this niche market, which is expected to produce growth rates of up to 10% annually in the coming years. Thus, there is a niche market for Chinese and foreign MNCs to target in this segment.
Over-the-counter Medicine in China
China's OTC medicine market remains one of the most attractive in the APAC region with a significant market share. However, government focus on improving access to prescription medicines will reduce demand for OTC drugs leading to an erosion of the domestic market share over the long term.
As OTCs make up between 30% and 40% of total industry revenue in developed countries, inexpensive OTC production in China for global markets is still expected to offer significant opportunities in future years.
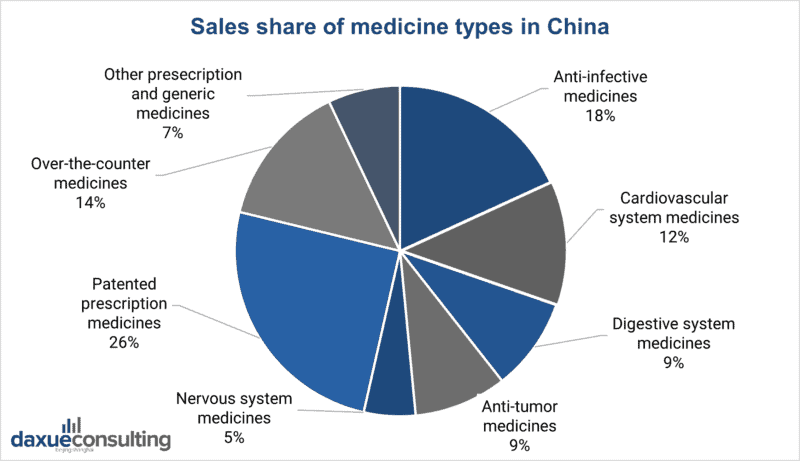 Source: IBISWorld. Designed by daxue consulting. The sales share of medicine types in China
Source: IBISWorld. Designed by daxue consulting. The sales share of medicine types in China
Major Sales Channels for Pharmaceuticals in China
1. Urban and Rural Hospitals
Hospitals have historically been the biggest sales channel for pharmaceuticals in China. In 2021, 63.5% of all pharmaceuticals sold domestically were sold through public hospitals. An additional 9.6% were sold through private hospitals, private clinics, and rural clinics. The rising enrolment rate in health care insurances will result in more demand. However, other sales channels are becoming increasingly prevalent.
2. Drugstores
Through expansion and increased coverage, drugstores have become the second largest sales channel for pharmaceuticals in China. In 2021, 26.9 % of drugs were sold through drugstores.
3. Online Sales
In line with the quickly rising popularity of online shopping, the Chinese pharmaceutical e-commerce market has also started to grow before the COVID-19 pandemic, albeit a bit more slowly. Jianke, a Guangzhou-based company with 100 million customers and over 680,000 SKUs in 2018 was the leading pharmaceuticals e-commerce platform before the COVID-19 pandemic.
However, since the COVID-19 outbreak, many Chinese customers began buying medicine on food delivery platforms such as Meituan and Ele.me. This has eaten into Jianke's market significantly, since Ele.me and Meituan are able to deliver medicine within thirty minutes, compared to up to one to two days that Jianke took. 87% of the products on these platforms are Western manufactured or made pharmaceutic products.
The process to order medicine online in China are also quite simple. The additional step of confirming a prescription only adds a minor inconvenience.
 Source: daxue consulting. How to order medicine in China through food delivery platforms
Source: daxue consulting. How to order medicine in China through food delivery platforms
4. Exports
Exports will remain steady in the next few years as Chinese pharmaceutical sector matures and starts looking toward fulfilling domestic demand and expanding abroad.
China exports pharmaceutical products to over 160 countries and regions. Despite decoupling efforts, Chinese exports are seeing continued and robust growth due to the high demand for generic medicines. Export growth will be further influenced by multinational companies entering the Chinese market to lower production costs, domestic companies expanding to foreign markets, and the Chinese government's emphasis on exporting pharmaceuticals as a key driver for the industry.
Pharmaceutical Trade and Investment in China – Challenges and Opportunities
Despite the remarkable prosperity of the market, the Chinese pharmaceutical industry is facing some unique challenges.
Rising production costs due to stricter production standards and ecological regulations
Lower profit margins as new policy demand continuous drug price adjustments
Stricter market entry rules and price controls
Therefore, domestic pharmaceutical companies with higher R&D costs are generally less profitable, though new policies are working to break down this trend. Although there have been increases in the number of patented drugs in the pharmaceutical industry in China, patents have made relatively low contributions to industrial value. On top of this, IP held by Chinese firms is less competitive compared with those of foreign companies and most of the pharmaceutical enterprises in China still focus on generic drugs.
China Is One of the Most Attractive Pharmaceutical Investment Destinations
While the Chinese pharmaceutical market continues to face uncertainty on the back of challenges around drug pricing and the low levels of IP protection, China still presents one of the most attractive investment destinations in the APAC region. Underpinning this notion, China boasts a number of inherent advantages that makes it well positioned for continued FDI flows into the pharmaceutical sector. These include a rapidly aging population, well-established manufacturing industry for pharmaceuticals, and notable government commitment to the sector's development.
Entering the Chinese Pharmaceutical Market – Good News for Foreign MNCs
Specific areas showing considerable market opportunities include:
Patent drugs for Tier-3 hospitals
Drugs for life-threatening diseases such as cancer, lung and liver diseases
New drug research and development
Pharmaceutical manufacturing
Off-patent generic growth (as GQCE enforcement improves)
Strong medicine sales growth
Rural and suburban areas
Pharmaceutical drug distribution
Steps to Improve Regulation and Licensing
China has taken considerable steps to improve the domestic market environment, specifically in terms of regulation and licensing operations for international companies. In 2019, the Center for Drug Evaluation and the NMPA reformed the application process for INDs and NDAs, allowing for accelerated review timelines that will bring both domestic and foreign drugs to market faster than ever before.
Chinese regulators also eliminated the requirement to conduct additional clinical trials in China before drugs could be released, another regulatory prerequisite that had been slowing down the addition of new drugs to the Chinese market. Now, drugs that have been tested and approved for market in other countries will be fast-tracked for review without having to undergo China-specific testing.
Public Insurance Policies
In addition to a favorable regulatory atmosphere, China's public health insurance policies have brought high-quality healthcare within reach to the average citizen. Hence, it seems the government remains committed to the improvement of healthcare access, exemplified by the universal healthcare scheme's expansion to cover chronic diseases. Specifically, in a push to improve healthcare coverage, more than 100 Western medicines were added to public insurance policies in 2017, including AstraZeneca's Brilinta (ticagrelor), and cancer treatments like Roche's Herceptin (trastuzumab), MabThera/Rituxan (rituximab), Avastin (bevacizumab) and Tarceva (erlotinib). A further update in 2018 added more cancer treatments to the national reimbursement list.
More R&D
Most of the top 20 multinational pharmaceutical companies have been expanding their footprint and are setting up more R&D facilities through various enterprise structures. Chinese consumers prefer multinational firms' pharmaceutical products due to concerns over the quality of domestic drugs as China in the past has been a prolific source of counterfeit and defective medicines.
Lifestyle-related diseases, like diabetes in China, are a growing concern for the government. Driven by an aging population, changing lifestyles, and increasing urbanization, China's rising non-communicable disease burden will be a central factor in supporting pharmaceutical market growth. This, combined with various government incentives to improve access to treatment will progressively support a greater multinational drugmaker presence.
Manufacturing Pharmaceuticals in China – Supply Chain Opportunities and Challenges
During the COVID-19 pandemic, international reliance on China as a pharmaceutical manufacturing hub for global pharmaceutic demand has become acutely clear, with Chinese domestic lockdowns – especially in the Yangtze Delta area, China's main pharma production region – leading to global supply chain disruptions and international pharmaceutic shortages. While both China and India are the top producers of generics, China dominates global API production, such that an estimated 70% of all Indian drugs contain APIs made in China; similar figures can be seen for drugs produced in Japan or Europe. According to Optima insights, in 2019 China was the source of 40% of global APIs. This is mainly due to low-cost production opportunities in China with its seasoned, large manufacturing facilities, comparatively lower energy costs, and less rigid environmental policies regarding chemical pollution produced during API manufacturing, beating production costs from India by 20% and even more for Western competitors. Nowadays, many APIs are only produced in China, often by a single supplier.
Disclaimer: This article is excerpted from the content originally published by Daxue Consulting. The opinions in the article do not represent ChemLinked BaiPharm Portal.








