On March 26th, 2021, China's National Medical Products Administration (NMPA) released the 2020 National Report on Adverse Drug Reaction Monitoring.
In 2020, China's National Monitoring System of Adverse Drug Reaction (ADR) received a total number of 1.676 million reports of ADR events. Among them, 506,000 new and serious ADR events were reported. In the past two decades, the number of ADR events reported shows an upward trajectory year by year.
In terms of drug type, most of the ADR events reported in 2020 involved chemical drugs, with a proportion of 83%. Anti-infective agents, and drugs for cardiovascular system, tumor, and nervous system were the Top 4 chemical drugs that reported ADR.
 Adverse Drug Reaction Events Classified by Drug Type
Adverse Drug Reaction Events Classified by Drug Type
In terms of routes of administration, injection and oral administration took up 56.7% and 38.1% respectively of all reported ADR events. Intravenous injection caused an overwhelming 91.1% of adverse reaction of all injection administration ADR events.
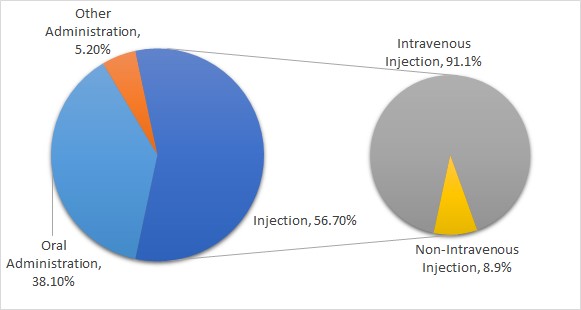 Adverse Drug Reaction Events Classified by Route of Administration
Adverse Drug Reaction Events Classified by Route of Administration
From the perspective of patient age group of all reported ADR events, middle-aged patient (45-64) accounted for 35.4%. Notably, the percentage of ADR events reported on children under 14 experienced a substantial decrease from 10.2% in 2019 to 7.7% in 2020. However, the percentage of ADR events reported on elders over the age 65 has been continuously rising in recent years.
 Adverse Drug Reaction Events Classified by Patient Age Group
Adverse Drug Reaction Events Classified by Patient Age Group








